Sodium fluoride
| Names | |
| Pronunciation | / ˌ s oʊ d I ə m e l ʊər aɪ d / [1] |
| IUPAC name Sodium fluoride | |
| Other names Flocid | |
| Identifiers | |
| Number of CAS |
|
| CHEBY |
|
| CHAMBL |
|
| ChemSpider |
|
| ECHA InfoCard | 100.028.789 |
| EU number |
|
| KEGG |
|
| PubChem C.I.D. |
|
| RTECS number |
|
| UNII |
|
| UN number | 1690 |
| CompTox Dashboard (EPA) |
|
InCHI
| |
| Characteristics | |
| Chemical formula | NaF |
| Molar mass | 41.988173 g/mol |
| Appearance | White to greenish solid |
| Smell | without smell |
| Density | 2.558 g/cm3 |
| Melting temperature | 993 °C (1819 °F, 1266 K) |
| Boiling point | 1704 °C (3099 °F, 1977 K) |
| Solubility in water | 36.4 g/l (0°C); 40.4 g/l (20 °C); 50.5 g/l (100 °C) [2] |
| Solubility | slightly soluble in ammonia, slightly soluble in alcohol, acetone, SO 2, dimethylformamide |
| Gas pressure | 1 mmHg Art. At 1077 C ° [3] |
| Magnetic susceptibility (χ) | −16.4 10 −6 cm 3 / mol |
| Refractive index ( n D) | 1,3252 |
| Structure | |
| Crystal structure | Cubic |
| Lattice constant | A = 462 pm |
| Molecular form | Octahedral |
| Thermochemistry | |
| Heat capacity ( C ) | 46.82 J/mol K |
| Standard molar entropy ( S o 298) | 51.3 J/mol K |
| Std formation enthalpy (Δ F H ⦵ 298 ) | -573.6 kJ/mol |
| Gibbs free energy (Δ f G ˚) | -543.3 kJ/mol |
| Pharmacology | |
| ATC code | A01AA01 (WHO) A12CD01 (WHO), V09IX06 (WHO) (18 F) |
| Dangers | |
| MSDS | [4] |
| GHS Pictograms | |
| GHS signal word | Danger |
| GHS Hazard Statements | H301, H315, H319, H335 [4] |
| NFPA 704 (fire diamond) | 0 3 0 |
| Flash point | Incombustible |
| Lethal dose or concentration (LD, LC): | |
| LD 50 (average dose) | 52–200 mg/kg (orally rats, mice, rabbits) [6] |
| NIOSH (US Health Exposure Limits): | |
| PEL (Permissible) | TWA 2.5 mg/m3 [5] |
| REL (recommended) | TWA 2.5 mg/m3 [5] |
| IDLH (Imminent Hazard) | 250 mg/m3 (as F) [5] |
| Related compounds | |
| Other anions | Sodium chloride Sodium bromide Sodium iodide Sodium astatide |
| Other cations | Lithium fluoride Potassium fluoride Rubidium fluoride Cesium fluoride France fluoride |
| Related compounds | TASF reagent |
| Unless otherwise stated, data is for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| N check (what is there?)YN | |
| Links to infoboxes | |
Sodium fluoride
(
NaF
) is an inorganic compound with the formula NaF. It is used in trace amounts in the fluoridation of drinking water, toothpaste, in metallurgy as a flux, and is also used in pesticides and rat poison. It is a colorless or white solid, readily soluble in water. It is a common source of fluoride in the production of pharmaceuticals and is used to prevent tooth decay.
In 2022, it was the 247th most commonly prescribed drug in the United States, with more than one million prescriptions. [7] [8]
Uses [edit]
Sodium fluoride is sold in tablets to prevent tooth decay.
Dental caries [edit]
See also: Fluoride therapy and water fluoridation
Fluoride salts are often added to municipal drinking water (as well as some foods in some countries) to promote dental health. Fluoride increases the strength of teeth by forming fluorapatite, a natural component of tooth enamel. [9] [10] [11] Although sodium fluoride is used for water fluoridation and is indeed the standard by which other water fluoridation compounds are measured, hexafluorosilicic acid (H 2 SiF 6 ) and its salt sodium hexafluorosilicate (Na 2 SiF 6 ) are more commonly used supplements in the USA [12].
Osteoporosis[edit]
Fluoride supplements have been studied extensively for the treatment of postmenopausal osteoporosis. This supplement does not seem to be effective; Although sodium fluoride increases bone density, it does not reduce the risk of fractures. [13] [14]
Medical imaging[edit]
In medical imaging, sodium fluoride labeled fluorine-18 (USP, sodium fluoride F18) is one of the oldest tracers used in positron emission tomography (PET), having been used since the 1960s. [15] Compared with traditional bone scintigraphy performed with gamma cameras or SPECT systems, PET provides greater sensitivity and spatial resolution. Fluorine-18 has a half-life of 110 minutes, which requires its immediate use after receipt; this logistical limitation has hindered its adoption in the face of more convenient radiopharmaceuticals labeled with technetium-99m. However, fluorine-18 is generally considered the best radiopharmaceutical for skeletal imaging. In particular, it has high and rapid absorption into bone tissue, accompanied by very rapid blood clearance, resulting in a high bone mass-to-background ratio in a short time. [16] Additionally, annihilation photons produced by the decay of 18 F have a high energy of 511 keV compared to the 140 keV photons of 99m Tc. [17]
Chemistry[edit]
Sodium fluoride has many specialty chemical uses in synthesis and mining metallurgy. It reacts with electrophilic chlorides including acyl chlorides, sulfur chlorides and phosphorus chloride. [18] Like other fluorides, sodium fluoride finds use in desilylation in organic synthesis. Sodium fluoride can be used to produce fluorocarbons via the Finkelstein reaction; The advantage of this process is that it is easy to perform on a small scale, but is rarely used on an industrial scale due to the availability of more efficient methods (e.g. electrofluorination, Fowler's process).
Other uses[edit]
Sodium fluoride is used as a cleaning agent (such as "wash sour"). [19]
More than a century ago, sodium fluoride was used as a stomach poison for plant-eating insects. [20] Inorganic fluorides such as fluorosilicates and complex magnesium ions sodium fluoride such as magnesium fluorophosphate. They inhibit enzymes such as enolase that require Mg 2+ as a prosthetic group. Thus, fluoride poisoning prevents the transfer of phosphates in oxidative metabolism. [21]
The manufacturing process of products containing fluoride
The manufacturing process for fluoride products is similar to those used to manufacture other oral care formulations. Since most fluoride compounds used in hygiene and treatment are water-soluble, these products are relatively easy to manufacture.
The production process involves simply mixing the ingredients. It does not require the inclusion of any special solvents, reagents or emulsification technology. Large batches of products can be produced in stainless steel tanks. The first step is mixing water with glycerin. This is the basis of most products. Other ingredients are added sequentially. Depending on the type of final product, heating and cooling of the solutions may be required to dissolve the components.
At the end of the process, pH regulators are introduced. They ensure that the product has the correct balance of acid base. Flavorings are added at the end of the operation as they are heat sensitive. Once all processes are completed, the formulation is tested for pH, weight percent solids, and fluoride concentration. Next, the product is bottled into individual bottles with sealed caps. Products packaged in test tubes or with foaming dispensers require more complex sealing mechanisms during packaging.
Security[edit]
See also: Fluoride poisoning
The lethal dose for a 70 kg (154 lb) person is estimated to be 5–10 g.[19]
Fluorides, especially aqueous solutions of sodium fluoride, are quickly and quite intensively absorbed by the human body. [22]
Fluorides interfere with electron transport and calcium metabolism. Calcium is necessary to maintain cardiac membrane potential and regulate coagulation. Large amounts of fluoride salts or hydrofluoric acid can lead to fatal arrhythmia due to profound hypocalcemia. Chronic overabsorption can cause bone hardening, ligament calcification, and dental deposits. Fluoride may cause irritation or corrosion to the eyes, skin, and nasal membranes. [23]
Sodium fluoride is classified as toxic both when inhaled (dust or aerosol) and when taken orally. [24] In high enough doses, it has been shown to affect the heart and circulatory system. In the case of occupational exposure, the Occupational Safety and Health Administration and the National Institute for Occupational Safety and Health have set exposure limits at 2.5 mg/m3 over an eight-hour time-weighted average. [25]
At higher doses used to treat osteoporosis, plain sodium fluoride can cause leg pain and incomplete stress fractures when doses are too high; it also irritates the stomach, sometimes so much that it causes ulcers. Slow-release and enteric-coated versions of sodium fluoride do not have any significant gastric side effects and have milder and less frequent bone complications. [26] At lower doses used for water fluoridation, the only obvious side effect is dental fluorosis, which can change the appearance of children's teeth as they develop; these are generally modest impacts and are unlikely to have any real impact on aesthetics or public health. [27] Chronic ingestion of 1 ppm fluoride in drinking water can cause stained teeth (fluorosis), and exposure to 1.7 ppm causes staining in 30–50% of patients. [22]
About caries and the fight against it with fluoride
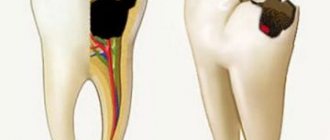
There are three main factors that influence the development of dental caries: tissue susceptibility, the presence of an infectious strain of bacteria (Streptococcus mutans), excess sugar and other nutrients that stimulate bacterial growth. As pathogenic bacteria grow, an acidic byproduct is produced that can dissolve the minerals in the enamel. As a result, the tooth is destroyed.
Dentists recommend the use of fluoride solutions for treating the oral cavity and drinking fluoridated water. It is thanks to fluoride that there is an improvement in the condition of teeth and a sharp reduction in caries. Similar improvements have been recorded in different studies and in almost all age groups. However, tooth decay remains the most common infectious disease, and fluoride treatment remains an important tool in the fight against dental and oral diseases.
Production[edit]
NaF is produced by neutralizing hydrofluoric acid or hexafluorosilicic acid (H 2 SiF 6 ), as by-products of the reaction of fluorapatite (Ca 5 (PO 4 ) 3 F) from phosphate rock, during the production of superphosphate fertilizer. Neutralizing agents include sodium hydroxide and sodium carbonate. Sometimes alcohols are used to precipitate NaF:
HF + NaOH → NaF + H 2 O
From solutions containing HF, sodium fluoride precipitates as the bifluoride salt of sodium bifluoride (NaHF 2 ). Heating the latter releases HF and gives NaF.
HF + NaF ⇌ NaHF 2
A 1986 report estimated the world's annual NaF consumption at several million tons. [19]
What is fluoride and how does it work?
Fluoride is a form of elemental fluorine, which is normally a toxic gas. Fluoride's chemical reactions with other compounds, such as tin, give it unique properties that increase the element's effectiveness in treating the oral cavity.
Once in the mouth, fluoride is diluted by saliva, depositing as a bacterial plaque on the surface of the teeth. In this state, it directly inhibits the growth of bacteria, so less acid is produced in the mouth. The fluoride stored in the plaque is then released when the bacteria produce enough acid to reduce the acid-base balance in the mouth. When this happens, fluoride diffuses into the tooth through tiny pores in the enamel. Fluoride ions replace the hydroxyl ions in the hydroxyapatite crystals that make up tooth enamel and form a new compound called fluorapatite. This compound is almost insoluble in the acids produced by bacteria in the mouth, so it helps protect teeth from decay.
Links[edit]
- ↑
Wells, John C. (2008),
Longman Pronunciation Dictionary
(3rd ed.), Longman, pp. 313 and 755, ISBN 9781405881180. According to this source, the alternative pronunciation of the second word /e l ɔːr aɪ d/ and, in UK, as well as /ел¯uərаɪd/. - Haynes, William M., ed. (2011). CRC Handbook of Chemistry and Physics (92nd ed.). CRC Press. clause 5.194. ISBN 978-1439855119.
- Lewis, Hazardous Properties of RJ Sax Industrial Materials. 10th ed. Volumes 1–3 New York, NY: John Wiley & Sons Inc., 1999, pp. 3248
- ^ ab Sigma-Aldrich Co. , Sodium fluoride. Retrieved March 17, 2015.
- ^ abc NIOSH Pocket Guide to Chemical Hazards. "#0563" . National Institute of Occupational Safety and Health (NIOSH).
- Martel, B.; Cassidy, K. (2004), Chemical Risk Analysis: A Practical Guide
, Butterworth-Heinemann, p. 363, ISBN 978-1-903996-65-2 - "Top 300 2020". ClinCalc
. Retrieved April 11, 2022. - "Sodium Fluoride - Drug Use Statistics". ClinCalc
. Retrieved April 11, 2022. - ↑
Bourne, volume editor, Geoffrey H. (1986).
Dietary Research and Advice on Health and Disease
. Basel: Karger. item 153. ISBN. 978-3-8055-4341-5. - ↑
Jr, Cornelis Klein, Cornelius S. Hurlbut (1999).
A Manual of Mineralogy: (by James D. Dana)
(21st ed., Rev. Ed.). New York: J. Wiley. ISBN 978-0-471-31266-6. - Selwitz, Robert H; Ishmael, in my midst; Pitts, Nigel B. (January 2007). "Caries". Lancet
.
369
(9555):51–59. DOI: 10.1016/S0140-6736(07)60031-2. PMID 17208642. S2CID 204616785. - Division of Oral Health, National Center for Prevention Services, CDC (1993), Fluoridation Census 1992 (PDF), retrieved 2008-12-29. CS1 maint: multiple names: list of authors (link)
- Haguenauer, D; Welch, W; Shi, B; Tugwell, P; Wells, G. (2000). "Fluoride for the treatment of postmenopausal osteoporosis." Cochrane Database of Systematic Reviews
(4): CD002825. DOI: 10.1002/14651858.CD002825. PMID 11034769. - Westergaard, P; Jorgensen, N. R.; Schwarz, P; Mosekilde, L (March 2008). "Effect of Fluoride Treatment on Bone Mineral Density and Fracture Risk—A Meta-Analysis." Osteoporosis International
.
19
(3): 257–68. DOI: 10.1007/s00198-007-0437-6. PMID 17701094. S2CID 25890845. - Blau, Monte; Ganatra, Ramanik; Bender, Merrill A. (January 1972). "18F-fluoride for bone imaging." Seminars in Nuclear Medicine
.
2
(1): 31–37. DOI: 10.1016/S0001-2998 (72) 80005-9. PMID 5059349. - Ordonez, A.A.; DeMarco, Vice President; Klunk, M. H.; Pokkali, S.; Jain, S.K. (October 2015). "Imaging chronic tuberculous lesions using sodium [18F] fluoride positron emission tomography in mice". Molecular Imaging and Biology
.
17
(5):609–614. DOI: 10.1007/s11307-015-0836-6. PMC 4561601. PMID 25750032. - Grant, F.O.; Fahey, F. H.; Packard, AB; Davis, R. T.; Alavi, A.; Treves, S. T. (December 12, 2007). "18F-Fluoride Skeletal PET: Applying New Technology to an Old Tracer". Journal of Nuclear Medicine
.
49
(1): 68–78. DOI: 10.2967/jnumed.106.037200. PMID 18077529. - Halpern, D.F. (2001), "Sodium fluoride", Encyclopedia of Reagents for Organic Synthesis
, John Wiley & Sons, DOI: 10.1002/047084289X.rs071, ISBN 978-0471936237 - ^ abc Aigueperse, Jean; Mollard, Paul; Devillier, Didier; Chemla, Marius; Faron, Robert; Romano, Rene; Couer, Jean-Pierre (2000). "Inorganic fluorine compounds." Ullman Encyclopedia of Industrial Chemistry
. Weinheim: Wiley-VCH. DOI: 10.1002/14356007.a11_307. - House, James E.; House, Kathleen A. (2015/09/10). Descriptive Inorganic Chemistry. Academic press. item 397. ISBN. 978-0-12-802979-4.
- Metcalfe, Robert L. (2007), "Insect Control", Ullman Encyclopedia of Industrial Chemistry (7th ed.), Wiley, p. 9
- ^ ab Kapp, Robert (2005), "Fluoride", Encyclopedia of Toxicology
,
2
(2nd ed.), Elsevier, pp. 343–346. - Greene Shepherd (2005), "Fluoride", Encyclopedia of Toxicology
,
2
(2nd ed.), Elsevier, pp. 342–343 - NaF Data Safety Data Sheet. hazar.com
- CDC - NIOSH Pocket Guide to Chemical Hazards
- Murray TM, Ste-Marie LG. Prevention and treatment of osteoporosis: consensus statements of the Scientific Advisory Board of the Canadian Osteoporosis Society. 7. Fluoride therapy for osteoporosis. CMAJ
. 1996. 155 (7): 949–54. PMID 8837545. - National Health and Medical Research Council (Australia). A systematic review of the effectiveness and safety of fluoridation
[PDF].
2007. ISBN 1-86496-415-4. Summary: Yeung CA. A systematic review of the effectiveness and safety of fluoridation. Proven dent
. 2008. 9 (2): 39–43. DOI: 10.1038/sj.ebd.6400578. PMID 18584000. - Wells, A.F. (1984), Structural Inorganic Chemistry
, Oxford: Clarendon Press, ISBN 978-0-19-855370-0 - "Chemical and Physical Information", Toxicological Profile of Fluorides, Hydrogen Fluoride, and Fluorine (PDF), Agency for Toxic Substances and Disease Registry (ATDSR), September 2003, page 187, accessed 11/01/2008
- Handbook of Minerals (PDF), mineral data publication, 2005.
How relevant will fluoride products be in the future?
In the future, it is possible that products containing fluorides will have serious competitors.
But today they still remain an important tool in the fight against infectious diseases of the teeth and oral cavity. New technological advances are appearing, in particular, for example, British researchers have discovered a new type of anti-caries agent that stops the development of caries in 3 months. The new ingredient is a protein fragment called p1025 peptide. It attaches to the surfaces of teeth and neutralizes pathogenic bacteria. It is likely that such developments and discoveries will one day help create fluoride-free products for the prevention and treatment of caries. Moscow metro station Zvezdnaya, Danube Avenue, 23
Interaction
The instructions indicate that calcium salts bind fluorine-containing compounds in the gastrointestinal tract. In addition, drugs based on aluminum or magnesium have a similar effect. Medicines that have enveloping properties also interfere with the absorption of fluoride, so they must be taken separately. Vitamins A and D can enhance the effect of sodium fluoride.
Treatment with sodium fluoride is effective in the fight against tooth decay and osteoporosis, but you should always follow your doctor's recommendations and periodically take blood tests to prevent possible side effects.
How to choose the right dosage
Your doctor should determine the dose. Instructions for the use of sodium fluoride indicate that for osteoporosis, the course of treatment should be long and last at least a year. The approximate dose for an adult is 0.015–0.02 grams 3 times a day. For preventive purposes, it is enough to take 1 tablet of sodium fluoride once a day. For the treatment of caries in a child, the dose ranges from 0.0011 to 0.0022 grams per day, but it must be agreed with the doctor. For children (age 16 years and older), mouth rinses with a 0.05–0.2% sodium fluoride solution are best.
Receipt
In industry, sodium fluoride is obtained by alkaline hydrolysis of hexafluorosilicates:
The degree of hydrolysis is small, since the constant of the last reaction is pK = 10.8.
Adds HF to form sodium difluorohydrate:
With an excess of HF, higher sodium hydrofluorides are formed:
compounds for n = 1÷4 are known.
Strong non-volatile acids destroy sodium fluoride:
Saturated lithium hydroxide, due to the poor solubility of lithium fluoride, destroys sodium fluoride:
Forms hexafluorosilicates and hexafluoroaluminates:
Melted sodium fluoride is an electrolyte, therefore it can be decomposed by electrolysis into elements:
When is sodium fluoride contraindicated?
The fluorine compound is an inorganic complex, and therefore requires timely disposal from the body. If the patient has impaired renal or liver function, taking the drug is not recommended. Sodium fluoride has the ability to increase the acidity of gastric juice, which is undesirable for people suffering from peptic ulcers. Pregnancy and breastfeeding are also factors that are incompatible with fluoride-based medications. Sodium fluoride for children with caries is a necessary component of treatment, but it must be carried out under the supervision of the attending physician.